
06.06.23
Bardusch AG supports customers in validating their cleanroom garments according to GMP Annex 1 requirements
Mastering GMP Annex 1 requirements: bardusch AG's expertise in cleanroom textiles and reliable services. In our interview with Patrick Wenger, branch manager of the cleanroom laundry in Selzach, you will learn how bardusch meets the new requirements of GMP Annex 1 while offering customized solutions for your cleanroom processes. In doing so, bardusch relies on individual consulting, comprehensive cleanroom services and the highest quality standards, offering customers real added value for their demanding cleanroom processes.

Patrick, GMP Annex 1 comes into force on August 25, 2023, and brings new requirements for the manufacture of sterile drugs and other sensitive products. These new requirements also relate to cleanroom garments to ensure sterility and particle-free production. How do we at bardusch AG ensure that we meet these requirements?
At bardusch, we have prepared intensively for the new requirements of GMP Annex 1. We meet all the specifications, especially with regard to the validation of cleanroom garments. Our new cleanroom laundry in Selzach is equipped with state-of-the-art infrastructure according to ISO 14644, and our quality and environmental management system is certified according to ISO 9001:2015 and ISO 14001:2015. The RABC system (Risk Analysis and Biocontamination Control System) meets the requirements of EN 14065:2016.
How do we support our clientele in validating their individual cleanroom garments?
We advise our clientele personally on the selection of the optimal equipment and take time to understand their specific requirements. We are able to provide and validate customized textile solutions according to the specific requirements of each customer. Our experts help to optimize cleanroom processes effectively and according to standard requirements.
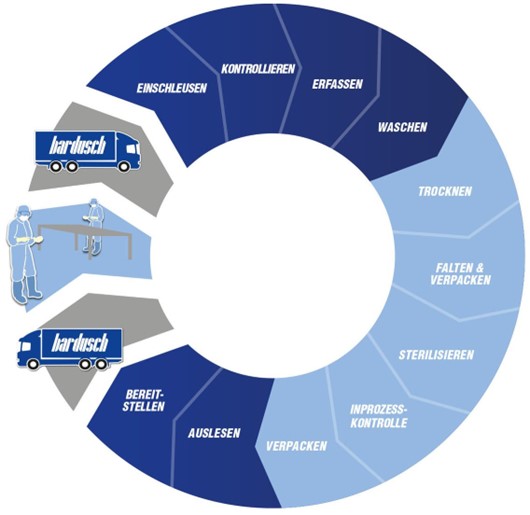
Could you tell more about the service cycle and quality controls that bardusch offers?
Our service cycle includes the collection, decontamination, sterilization and on-time delivery of reprocessed textiles. We ensure that our customers have the textiles they need on time and in the right quantities. Regular quality controls, 100% batch traceability and complete documentation are part of our standard and ensure compliance with GMP Annex 1 requirements.
What financing options does bardusch offer for cleanroom clothing and services?
We offer customer-oriented leasing price models that allow predictable monthly costs for perfectly refurbished textiles without tying up capital.
What makes bardusch unique in the field of cleanroom textiles and services?
We attach great importance to providing our customers with continuous support at every stage of the process. Our dedicated team is always available and ready to respond to our customers’ needs and requirements.
We guarantee 100% Swiss production under the “Swiss washed” label and have a nationwide presence in Switzerland, which enables us to always be close to our customers and to react quickly to changing situations. Our international network also ensures a reliable supply of cleanroom textiles, even in exceptional situations.

Finally, what advantages do customers have when they choose bardusch ?
Our clientele benefits from our comprehensive expertise and personal advice from our team of experts. Our cleanroom range and fully documented laundry cycle and quality control processes ensure that highly sensitive production processes are not contaminated by employees. By relying on bardusch, our clientele receives the highest quality, hygiene and efficiency.
How do interested parties best contact bardusch cleanroom laundry?
The best thing for them to do is visit our website or contact us. We look forward to supporting many interested parties with their cleanroom requirements.
For more information, click here: Cleanroom Laundry Service. (bardusch.com)

